Allergenic components of the mRNA‐1273 vaccine for COVID‐19: possible involvement of polyethylene glycol and IgG‐mediated complement activation
Allergenic components of the mRNA‐1273 vaccine for COVID‐19: possible involvement of polyethylene glycol and IgG‐mediated complement activation
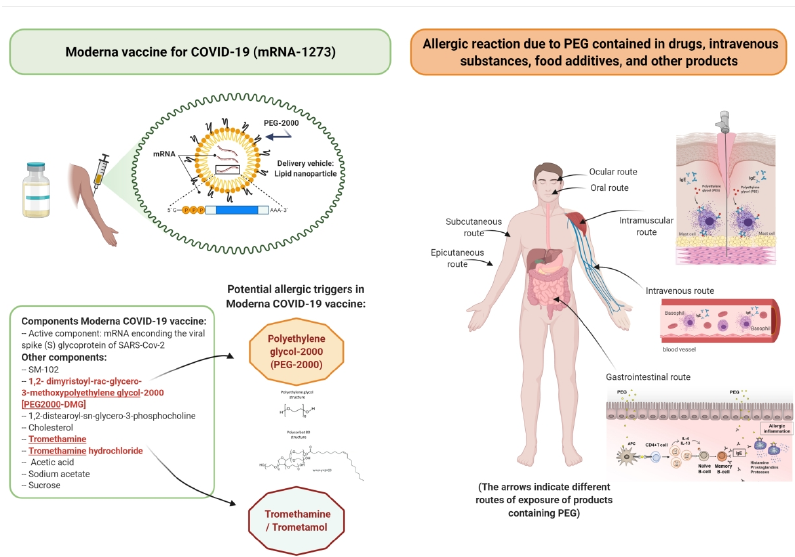
Following the emergency use authorization of the mRNA‐1273 vaccine on the 18thof December 2020,two mRNA vaccines are in current use for the prevention of coronavirus disease 2019 (COVID‐19). For both mRNA vaccines, the phase IIIpivotal trials excluded individuals with a history of allergy to vaccine components.